|
12/16/2023 0 Comments Fission reaction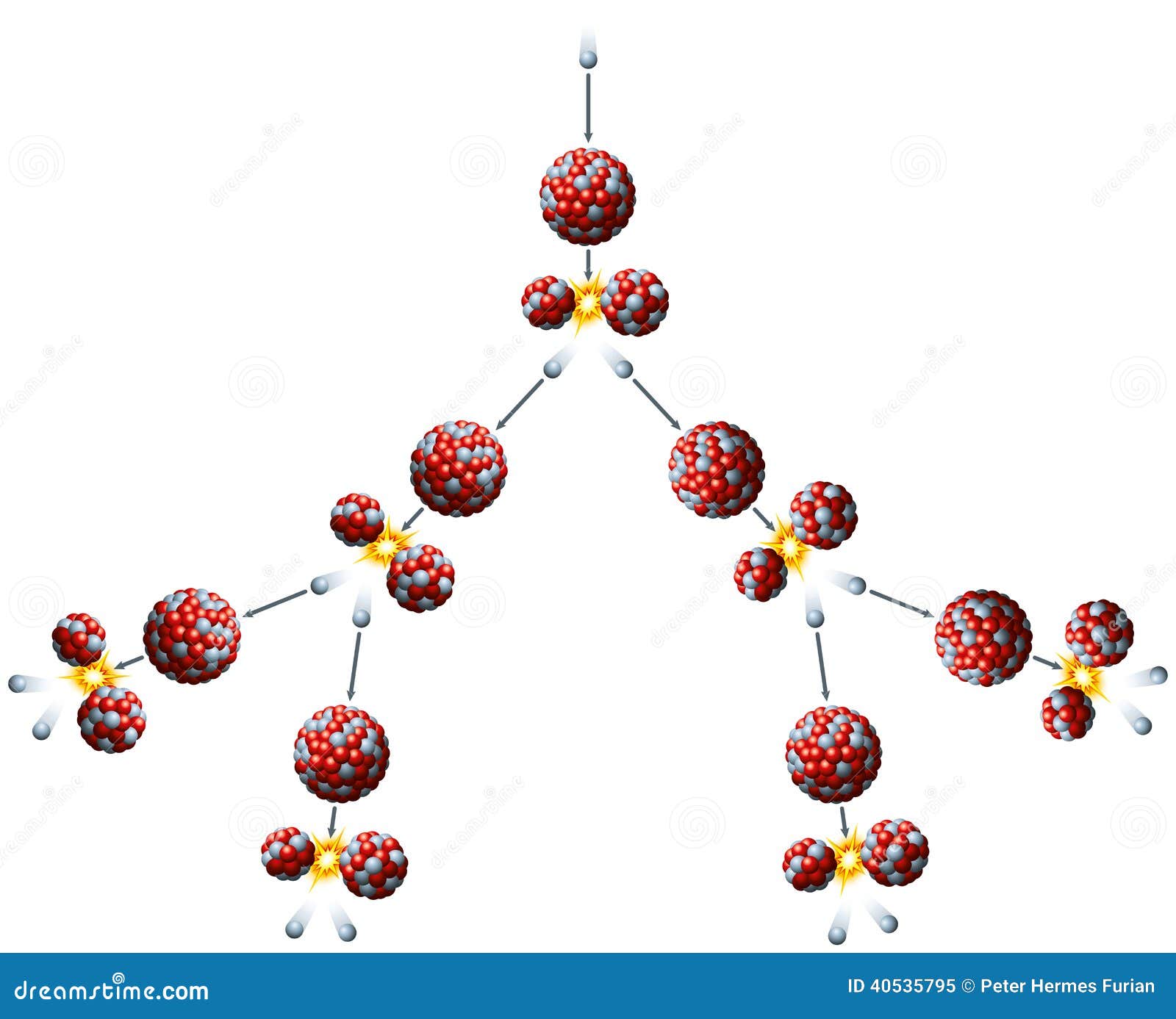
You only get about 200 MeV from the fission of a single atom of U-235. Nuclear fission of heavy elements was discovered on Monday 19 December 1938, by German chemist. The fission process often produces gamma photons, and releases a very large amount of energy even by the energetic standards of radioactive decay. Has anyone tried to balance the left hand side with the right hand side? What's the atomic number of plutonium? Of strontium?Ī gamma ray with an energy of 130,000 MeV is an extraordinary event. Nuclear fission is a reaction in which the nucleus of an atom splits into two or more smaller nuclei. Otherwise, a lot of time is wasted in going back and forth trying to make sense of what is being asked. I thought because the energy of the gamma ray is the only unknown then Q = energy of the gamma ray = 130520 MeVīecause the energy of the gamma ray is the amount of energy that has to be provided for the reaction of Pu-239 + gamma -> Sr-92 + He-4 + 3n to occur, this means gamma = -130520 MeVThis is why the PF HW forum rules ask posters to include the full question when filling out the HW template. I'm pretty sure that means Pu-239 + gamma -> Sr-92 + He-4 + 3n This occurs when neutrons collide with protons in water molecules, which provide a coolant and a moderator. The key step is moderation of fast neutrons produced by fission. A gamma ray with 130520 MeV must be provided to split a Pu-239 atom into Sr-92 + He-4 + 3nĬomplete the following fission reaction, and then calculate the Q-value for the reaction: Pu-239 + gamma -> Sr-92 + ? + 3n Nuclear fission reactions can sustain a nuclear controlled chain reaction and will produce both large quantities of thermal energy and neutrons. And why should I use the atomic mass of He-4 in this calculation instead of the atomic mass of the alpha particle?Ģ) Is Q negative because it represents the amount of energy which must be provided for the reaction to occur? I.e. This neutron is absorbed, turning the uranium-235 atom into an. The fission of 1 g of uranium or plutonium per day liberates about 1 MW. Thus, 3.1 ´ 10 10 fissions per second produce 1 W of thermal power. One fission event results in the release of about 200 MeV of energy, or about 3.2 ´ 10 -11 watt-seconds. They will define chain reaction in their own words after watching a demonstration using. Nuclear reactions liberate a large amount of energy compared to chemical reactions. The fission process often produces both free neutrons and gamma photons. Why is this? I know He-4 is missing it's two electrons, therefore it's mass is the equivalent of the alpha particle, but how come He-4 atomic mass ≠ alpha particle. Nuclear Fission Reaction To start the reaction above, you first have to take a sample of uranium-235 and fire a neutron at it. They will see an example of fission using balloons and marbles. In physics, nuclear fission is a nuclear reaction or radioactive decay process where the nucleus of an atom splits into smaller (and lighter) nuclei. = - 130520 MeV (correct to 5 significant figures)ġ) I've seen examples where He-4 = 4.0026032497 is used in this calculation instead of the Alpha Particle = 4.00150618. Q-Value = ? (Answer in MeV, correct to 5 significant figures) Fission Reaction: Pu-239 + gamma -> Sr-92 + ? + 3n
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
